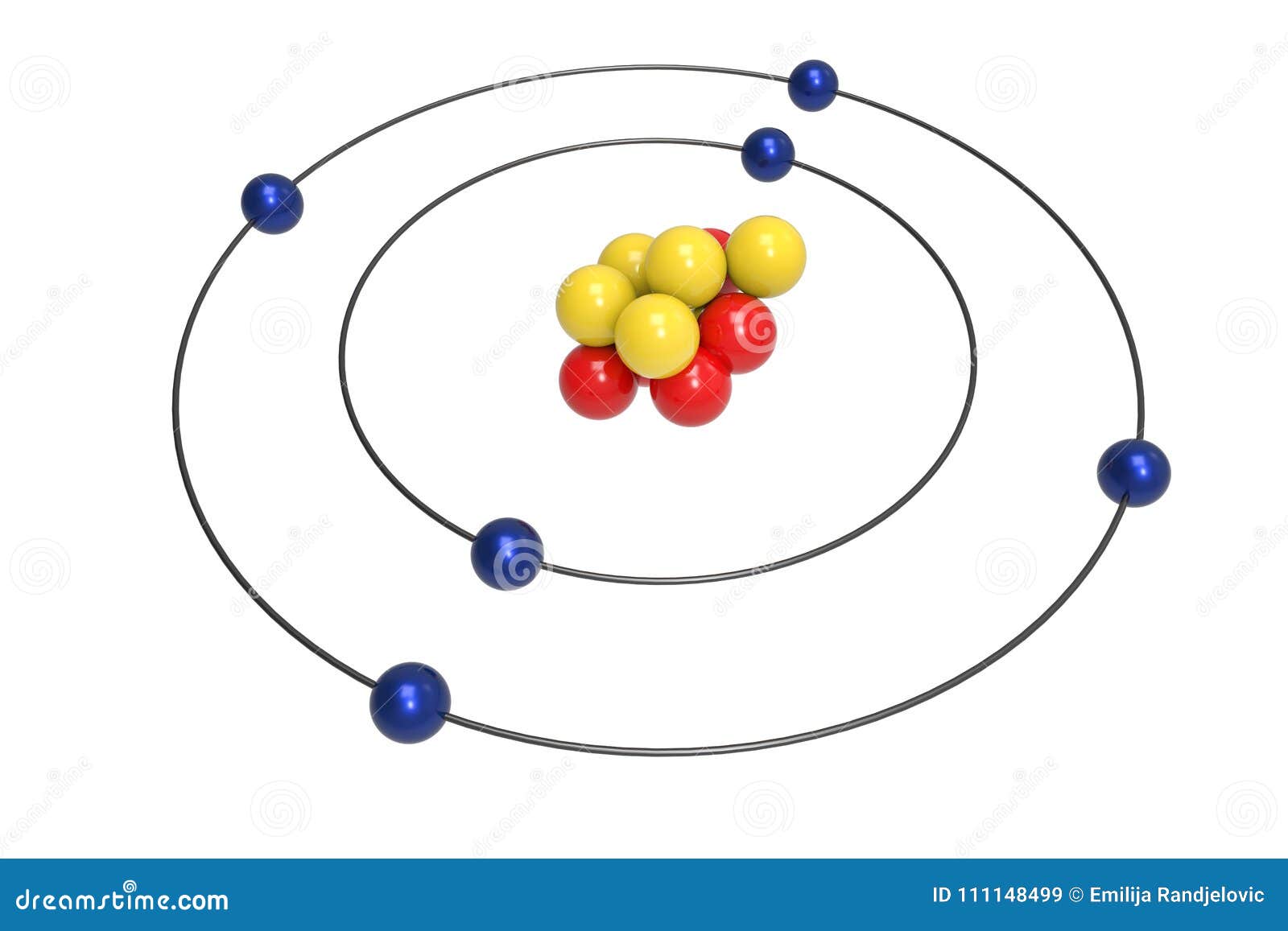 
The bigger the orbit is, the bigger the energy is. The energy of the electron is different on different orbits. On each orbit the electron has some defined energy. The second postulate says that in an atom an electron can change orbits. But he was of the opinion that to properly understand the nature of the atom one has to accept his idea. The postulate was in complete disagreement with other theories, and especially with the Maxwell theory of electromagnetism. Bohr formulated the postulate ad hoc. Electrons moving on them don’t lose energy for radiation. The first one says that electrons can’t move on unlimited orbits around the nucleus. Let’s now think what each postulate means: 
The laws of mechanics describe the dynamic equilibrium of electrons in stationery states but do not describe the situation of the electron transition from one stationery orbit to another.Where h is the Planck constant, E n and E l are the energies in the two stationary states. The radiation emitted during such transition is homogeneous and its frequency is given by the formula: Each emission or absorption of radiation energy represents the electron transition from one stationery orbit to another.There are some orbits called by him the stationery ones, where the moving electrons don’t emit energy.Just like Rutherford he assumed that electrons rotate around the nucleus. In 1913 Niels Henrik Bohr published his new theory of the atoms constitution.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
